It is the simplest PNH. It occurs as a white crystalline solid with a characteristic strong odor. It is obtained from either coal tar or petroleum distillation. It is used as dyes, plastics, lubricants, and explosives. When high levels of naphthalene vapors are inhaled, it could cause nausea, vomiting, and headache.
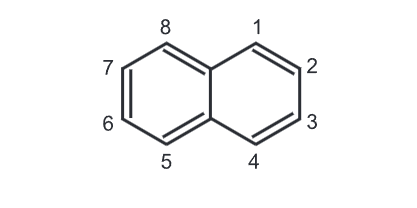
Chemical Reaction of Naphthalene
(i) Reduction: Naphthalene can be partially or fully reduced under different reagent conditions.
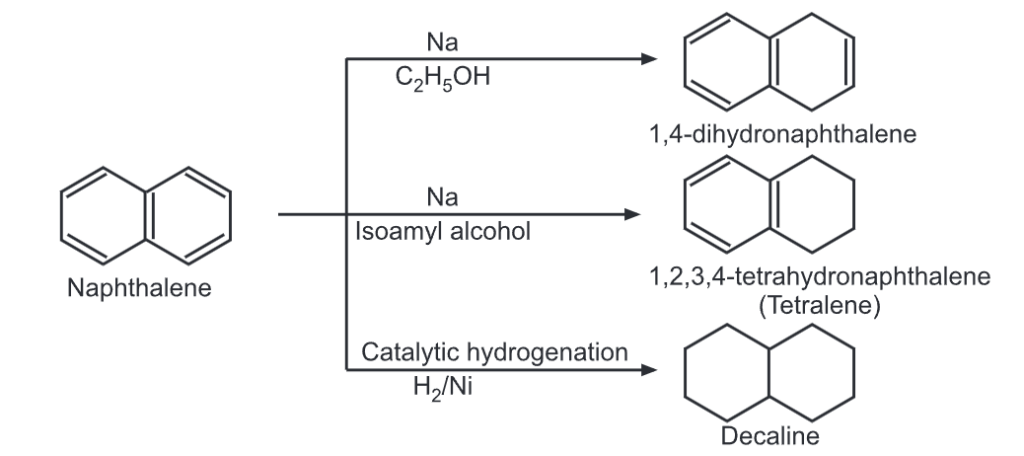
(ii) Oxidation: Naphthalene when oxidized under four different conditions, gives four different oxidized products.
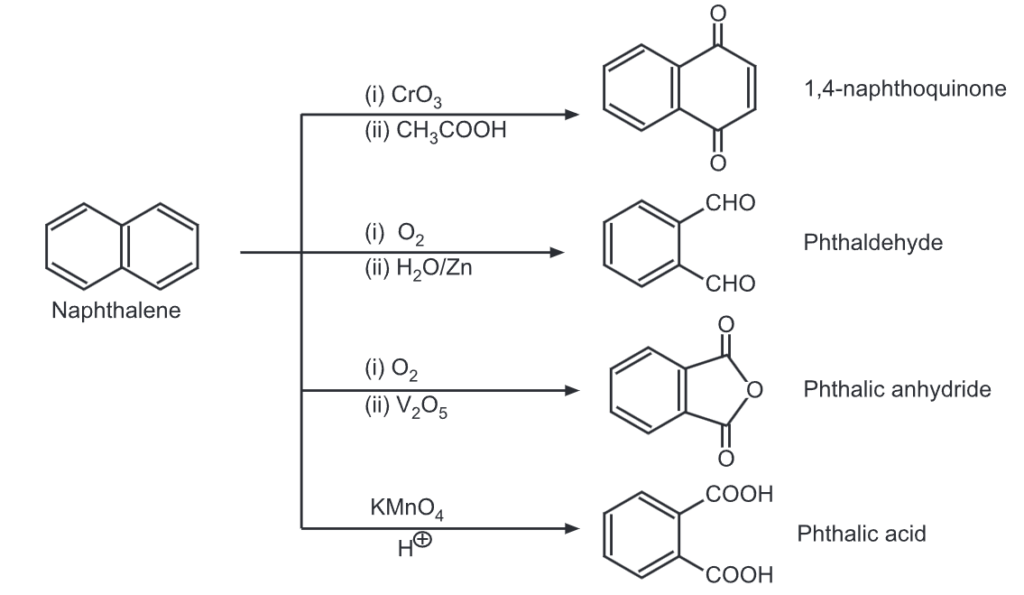
(iii) Chlorination:
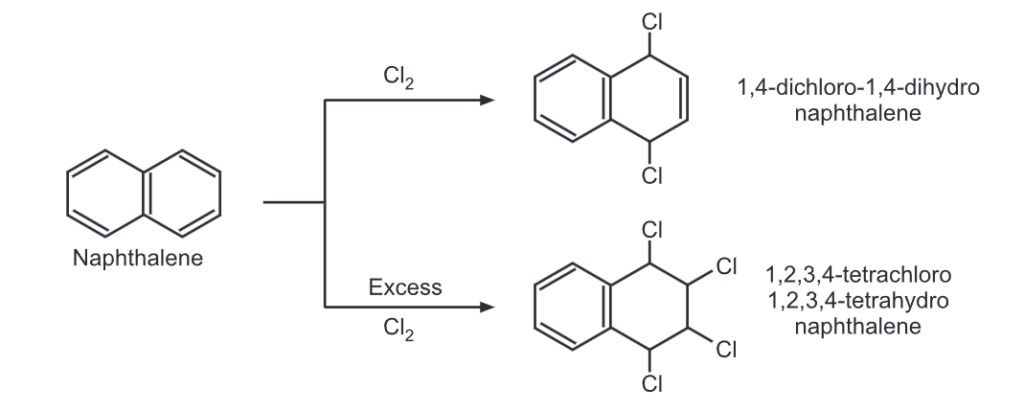
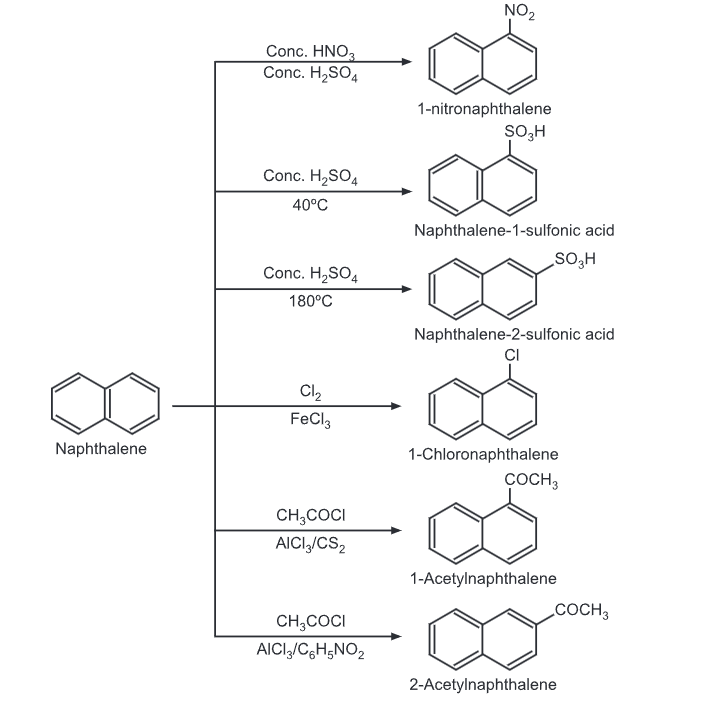
Electrophilic Substitution Reaction of Naphthalene
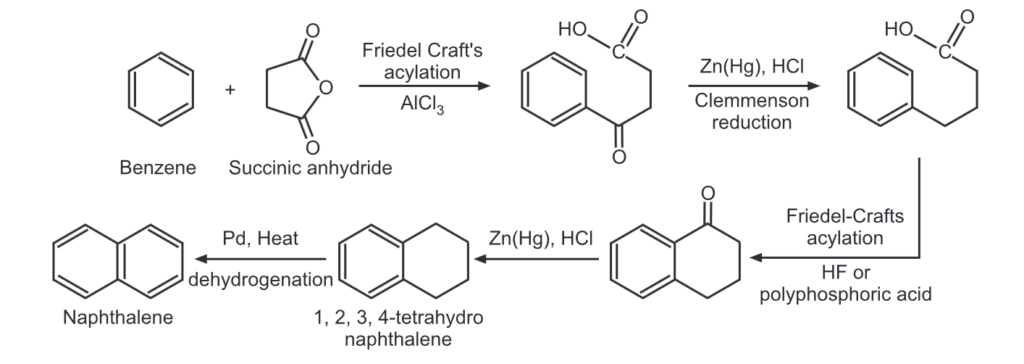
Synthesis of Naphthalene
Make sure you also check our other amazing Article on : Reaction of Diphenylmethane
