Principle
In a vacuum crystallizer, supersaturation is obtained by adiabatic evaporative cooling. When the warm saturated solution is introduced into the crystallizer, due to high vacuum the solution undergoes flashing. A part of the solvent gets evaporated, thereby causing the cooling of the solution. From the resulting supersaturation, crystals are produced.
Construction
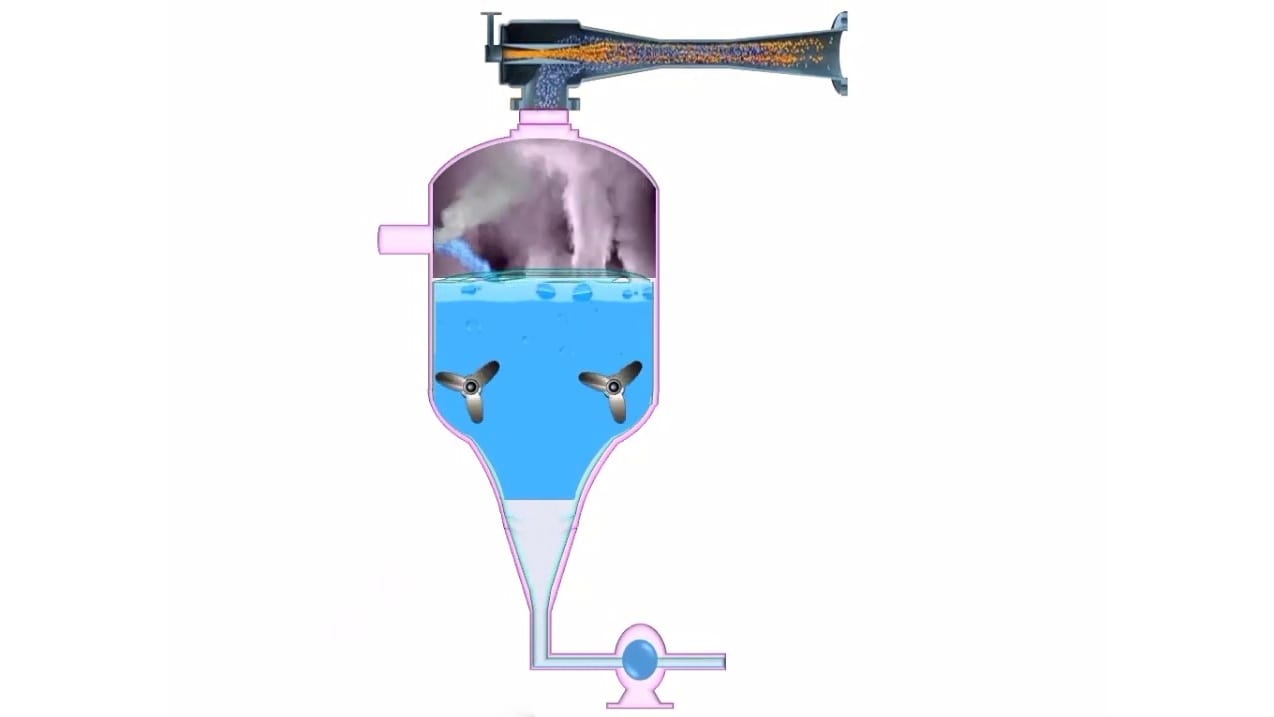
The construction of a vacuum crystallizer is shown in Figure. A vacuum crystallizer is a cylindrical body with a conical bottom. A condenser is attached to the crystallizer with a vacuum pump in between. The bottom of the crystallizer is attached to a discharge pipe. Internally, the body of the crystallizer can be lined with acid-resistant material such as lead or rubber. Two propellers are placed above the discharge pipe to prevent a short circuit of the feed (to the discharge pipe).

Working
A high vacuum is created using a vacuum pump. The vacuum so created must correspond to the boiling point of the solution. but lower than the feed temperature. A hot saturated solution is fed into the crystallizer at a convenient point. Solution undergoes flashing, which results in evaporation of the solvent. This process is allowed adiabatically so that the crystallizer body is cooled. The resultant cooling causes supersaturation and crystallization. The evaporation of the solvent enhances the yield. Flashing of the solution in the crystallizer leads to ebullition, which keeps the crystals in suspension until they become large enough to fall into the discharge pipe. The propellers mix the contents thoroughly and prevent the contents from reaching the discharge pipe without flashing. With the help of a pump, the product is collected and subjected to filtration or centrifugation to obtain crystals. The filtrate returns to the feed.
Uses: It is suitable for thermolabile substances, due to low-temperature conditions.
Advantages:
- The vacuum crystallizer is very simple without many moving parts.
- Corrosive materials can be used, as the inner surface can be made acid-resistant.
- It can be constructed as large size as desired.
- It can be operated either batch-wise or continuously.
Make sure you also check our other amazing Article on : Falling Film Molecular Still