Clean rooms are maintained particulate-free air through the use of either HEPA or ULPA filters based on laminar or turbulent air flow principles. Laminar or unidirectional, air flow systems direct filtered air downward in a constant stream. Laminar air flow systems are typically employed across 100% of the ceiling to maintain constant and unidirectional flow. Clean Area Classification below the equation. The maximum permitted concentration of particles Cn for each considered particle size D is determined from the equation:
Cn = (0.1/D)2.08 × 10N
where,
Cn = The maximum permitted concentration (particles/m3 ) of airborne particles that are equal to or larger than the considered particle size.
N = The ISO classification number in the range from 1 to 9.
D = The considered particle size (µm).
Classification of Clean Area
The clean room classification standards FS 209E and ISO 14644-1 require specific particle count measurements and calculations to classify the cleanliness level. In the UK, British Standard 5295 is used to classify clean rooms. A clean area is classified into several grades which are listed in Table 1.1.
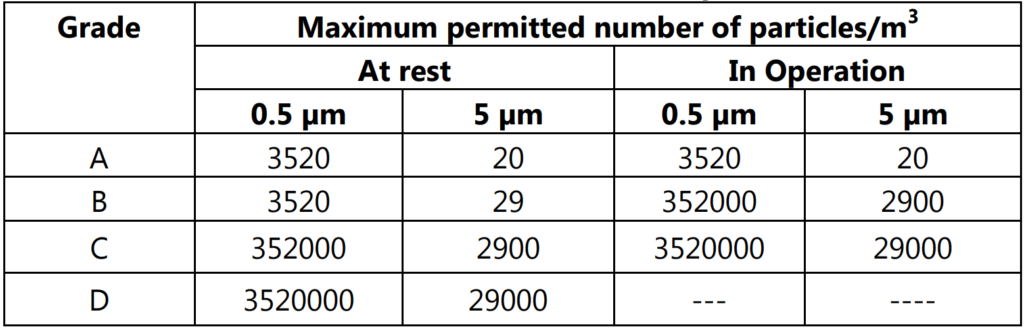
Table 4.2 indicates that for Grade A the airborne particle classification is ISO 4.8 dictated by the limit for particles ≥ 5.0 µm. For Grade B the airborne particle classification is ISO 5 for both particle sizes considered. For Grade C the airborne particle classification is ISO 7 and ISO 8, respectively and for Grade D the airborne particle classification is ISO 8.
Table 4.2 also summarizes the clean area air classifications and recommended action levels of microbiological quality in Table 1.2.
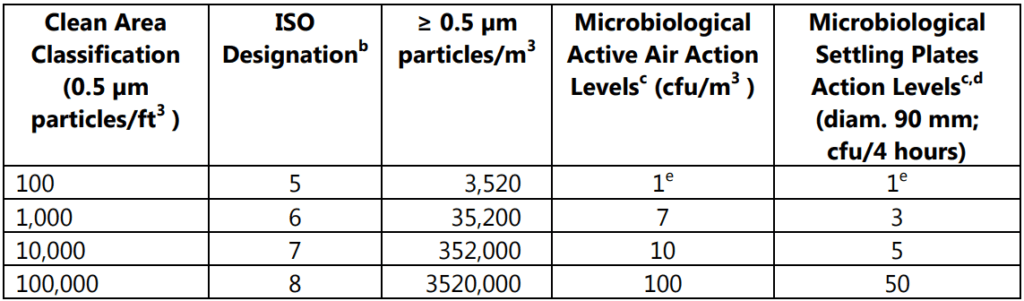
- a: All classifications are based on data measured in the vicinity of exposed materials/articles during periods of activity.
- b: ISO 14644-1 designation provides uniform particle concentration values for clean rooms in multiple industries. An ISO 5 particle concentration is equal to Class 100 and approximately equals EU Grade A.
- c: Values represent recommended levels of environmental quality. You may find it appropriate to establish alternate microbiological action levels due to the nature of the operation or method of analysis
- d: The additional use of settling plates is optional.
- e: Samples from Class 100 (ISO 5) environments should normally yield no microbiological contaminants.
Two clean areas are important to the quality of sterile drug products namely the critical area and the supporting clean area. The critical area is the area where the sterilized drug product, containers, and closures are exposed to environmental conditions that must be designed to maintain product sterility. The activities conducted in this area are aseptic connections, and sterile ingredient additions of sterile materials prior to and during filling and closing operations. This area is critical because an exposed product is vulnerable to contamination and will not be subsequently sterilized in its immediate container. To maintain product sterility, it is essential to maintain the proper quality of the environment in which aseptic operations are conducted.
Supporting clean areas contain many support areas that function as zones in which non-sterile components, formulated products, in-process materials, equipment, and container/closures are prepared, stored, and transferred. These environments are designed to control the microbiological content of articles and sterilized components.
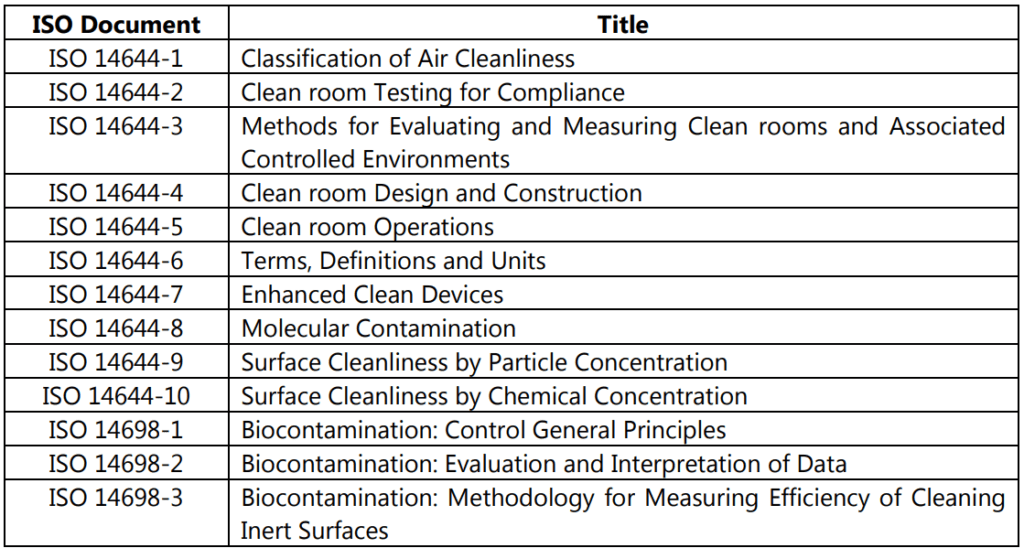
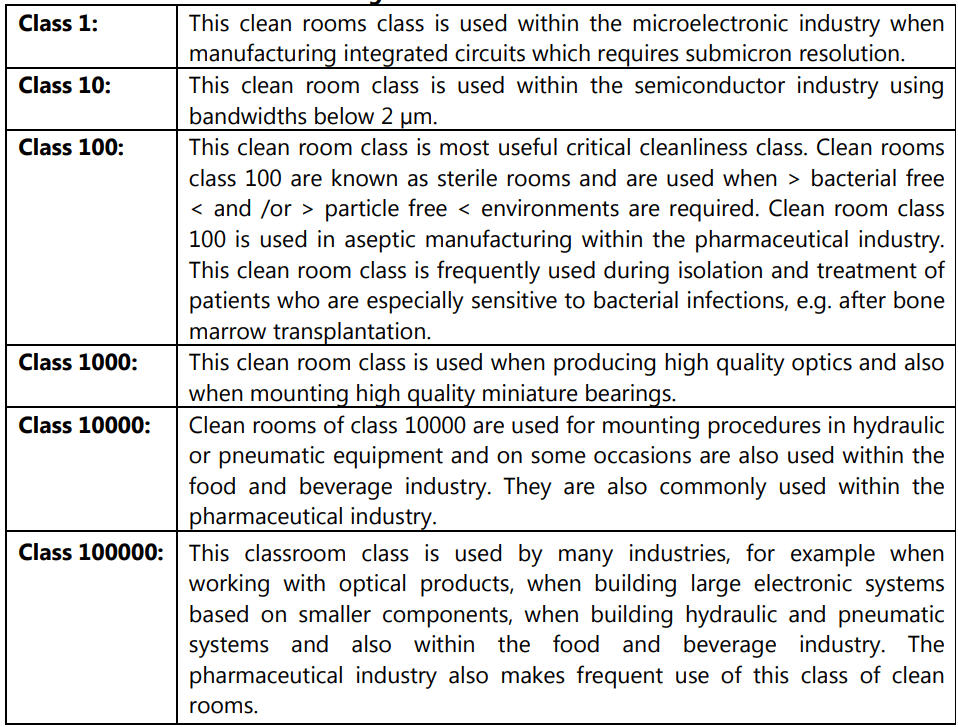
As per ISO 14644-1 and ISO 14644-2, eight other clean room standards documents exist and their meanings are tabulated in Table 1.3 and Table 1.4.
Make sure you also check our other amazing Article on : Buffered Isotonic Solutions
