Tablet coating is one of the oldest pharmaceutical processes. It involves the application of sugar or polymeric coat on the tablet. The advantage of coating tablets is taste masking, odor masking, physical and chemical protection, protecting the drug in the stomach, and controlling its release profile.
A summary of the rationale for tablet coatings is provided below:
- To protect the drug from degradation in the stomach (an enteric coating).
- To prevent drug-induced irritation at a specific site within the gastrointestinal tract, e.g. the stomach for non-steroidal anti-inflammatory drugs (NSAIDs) .
- To provide controlled release of the drug throughout the gastrointestinal tract.
- To target drug release to a specific site in the gastrointestinal tract, e.g. the delivery of drug to the colon for the treatment of inflammatory conditions.
- To mask the taste of drugs.
- To improve the appearance of the tablet.
General Description of Tablet Coating:
The main steps involved in the coating of tablets are as follows:
- The tablets (or granules) are placed within the coating apparatus and agitated.
- The coating solution is sprayed onto the surface of the tablets.
- Warm air is passed over the tablets to facilitate the removal of the solvent from the adsorbed layer of coating solution on the surface of the tablets.
- When the solvent has evaporated, the tablets will be coated with the solid component of the original coating solution.
Table of Contents
Coating Formation
Coating solutions are available in two main formulation types :
- Solutions
- Emulsions.
Coating solutions
Coating solutions contain the coating material (polymers or sugar), the coating solvent, and other excipients that are required to improve the performance of the tablet coating, e.g. colorants/opacifiers, plasticizers (to render the film flexible). The choice of the solvent/solvent blend is according to the physicochemical properties of the coating material (i.e. the compatibility of the material with the solvent); however, other considerations include the volatility and the flammability of the solvent. The concentration of the coating material within the solution is also a consideration. Increasing the concentration of coating material within the solvent will reduce the processing time; however, by increasing the concentration of material, the viscosity of the solution may be unacceptably high to achieve the correct spray properties during coating.
Coating emulsions
More recently emulsions have been developed as tablet coating systems. In these, the polymer is dissolved in a volatile organic phase (with plasticizer and colorants/opacifiers, as required) and this is emulsified within an external aqueous phase. The initial stage in the coating process involves the deposition and subsequent spreading of the atomized coating solution/emulsion on the surface of the tablet (or granule). To achieve a uniform surface distribution of the coating solution/emulsion on the tablet, consideration of the wetting properties of the solution/emulsion on the surface of the tablet is required. Following spreading, evaporation of the solvent initially enables coalescence of the organic droplets, and hence initial film formation on the surface of the tablet. As drying continues, the saturation solubility of the coating material in the solvent is exceeded and the solid coating is formed on the surface of the tablet. It should be noted that contact, spreading, droplet coalescence, and solvent evaporation occur almost instantaneously.
Tablet Coating in Practice
There are several designs of systems that are used in industrial practice to coat tablets (or granules).
Examples of these systems are:
Pan Coater
The pan coating system is generically composed of a metal pan (drum) into which the tablets are placed and that may be rotated at a range of speeds. The coating solution is sprayed onto the surface of the tablets within the pan whilst the drum is rotated. Simultaneously warm air is passed over the surface of the tablets to facilitate the evaporation of the solvent in which the coating material has been dissolved.
Control of the coating process is obtained by modifying the following parameters:
- The rotation rate of the drum/pan
- Airflow rate
- The temperature of the air
- The concentration of sugar/polymer within the coating solution/emulsion
More recently, pan coaters have been developed in which the pan is perforated (e.g. the Accela-Cota and Hi-Coater systems). In these systems, the warmed air is passed into the drum and through the tablet bed before being exhausted (with the solvent from the coating solution) via the perforated drum. In the Driacoater system, the drum is composed of a series of perforated fins (typically 8 per drum) from which the warmed air is provided. As the drum rotates, the tablets in the tablet bed are mixed by and collected on the fins before being suspended in the warmed air. The tablets are then dropped into the tablet bed and the process is repeated. The warmed air is then exited from the rear of the pan.
Air Suspension Coaters
Air suspension coaters are highly efficient coating systems in which the coating solution is sprayed onto tablets (or granules) that have been suspended in a positive (warmed) airflow. This ability simultaneously to suspend and coat tablets lead to high coating efficiency. Typically the tablets are initially suspended in the center of the chamber and then move to the periphery of the chamber before falling to the bottom, at which stage the process is continuously repeated. The coating solution is fed into the fluidization chamber (usually at the bottom of the chamber) as an atomized spray that has been generated either by the passage of the coating solution through a nozzle under high pressure or by the passage of the coating solution through a nozzle at low pressure, at which point the solution comes into contact with two high-pressure air streams.
Process Variables in Fluidized Air Coating
Several process factors control both the efficiency of the coating process and the quality of the formed coat. These are:
- Evaporation rate of the solvent.
- Fluidized air volume.
- Specific humidity.
- Coating sprays rate and duration.
Evaporation rate of the solvent: The rate of evaporation of the solvent directly affects both the quality (in particular the mechanical properties) of the tablet coating and the time required to form the tablet coating. Whilst it is important to process the coating in the minimum time, increasing the evaporation rate of the solvent decreases the time available for polymer-polymer interactions to occur. Therefore, if the solvent evaporation rate is too rapid, the mechanical properties of the films will be compromised due to the adverse effects on polymer interactions. Both the solvent vapor pressure and the process temperature affect the rate of evaporation of a solvent. Therefore, a low process temperature is normally employed for coating solutions/solvents containing a solvent of high vapor pressure, e.g. dichloromethane. Small fluctuations in processing temperature will have greater effects on the quality of the tablet coatings prepared using organic solvents than when processed using an aqueous coating solution.
Fluidized air volume: The fluidized air volume will affect both the velocity of the droplets of coating solutions/emulsions and their fluidized pattern within the coating chamber.
Specific humidity: It is important to control the specific humidity within the warmed air and hence in the coating chamber to ensure that the quality of the tablet coating is optimized. If the relative humidity in the coating chamber is high, evaporative cooling by the solvent may occur. This will, in turn, lower the temperature of the air to below the dew point, resulting in the condensation of water onto the tablet surface. This will interfere with the coating process, resulting in poor adhesion of hydrophobic coatings to the tablet surface and visual imperfections in the formed coating. Therefore, control (but not elimination) of the relative humidity within the coating process is required. The presence of humidity within the coating chamber may be useful in dispelling static electricity that may occur after the coating process has been completed.
Coating spray rate and duration: The coating spray rate is controlled within the coating process and is selected according to the solubility of the coating solvent in the air volume and the viscosity of the atomized droplets. It should be noted that excessive spray rates will produce coatings that exhibit poor adhesion to the tablet surface. Typically the coating process will involve several passes through the coating apparatus. Therefore one method by which the thickness of the coating on the tablet may be modified is to increase the time spent within the coating chamber. Alternatively, the concentration of coating material may be increased within the coating solution. The viscosity of the solution must be considered to ensure that the increased viscosity does not compromise the atomization process, and specifically the droplet size.
Problems Associated with Tablet Coatings
There are several problems associated with tablet coatings, including:
- Poor adhesion of the coating to the tablet;
- Tablet abrasion;
- Filling tablet markings;
- Rough surface;
- Formation of cracks in the coating; and
- Variations in the color of the coating.
Poor Adhesion of the Coating to the Tablet:
This phenomenon may be due to:
- High relative humidity within the coating chamber when coating tablets using an organic solvent system.
- High coating spray rate.
- The concentration of polymer in the coating solution/emulsion is too low.
- The temperature of the air is too low, resulting in a slow rate of solvent evaporation (particularly valid for coating systems that employ solvents of low vapor pressure, e.g. water).
- The Air fluidization rate or pan rotation rate is too slow.
- The tablet substrate has minimal curvature. Typically curved surfaces are easier to coat than flat surfaces.
Tablet Abrasion
The coating process involves exposing the tablets to shearing stresses that are generated as a result of collisions with other tablets and also with the walls of the coating chamber. This may result in damage to the tablet surface. This problem may occur due to:
- Inappropriate tablet hardness.
- Irregular tablet shape.
- The tablet bed is too heavy during coating.
- The speed of rotation of the pan or the air fluidization rate is excessive.
Inappropriate tablet hardness: Tablet hardness may be improved by increasing the compaction pressure or binder concentration (in wet granulation). Generally, the hardness of tablets produced by wet granulation is greater than by other methods and therefore the tablets produced by this method are generally suitable for coating.
Irregular tablet shape: Irregular tablet shapes are more prone to abrasion than regular tablet shapes. This problem may therefore be overcome by changing the tablet shape.
The tablet bed is too heavy during coating: This leads to increased tablet–tablet contact. To correct this problem the loading of tablets within the coating chamber is reduced.
The speed of rotation of the pan or the air fluidization rate is excessive: By reducing the speed of pan rotation or air fluidization rate, tablet–tablet contact is decreased.
Filling Tablet Markings
Manufacturers may wish to identify their product with a particular mark/name (performed by using a tablet punch that has been embossed with the specified mark). If the coating conditions are unsuitable, the coating will excessively deposit within the mark/name, and, in so doing, the marking will be partially obscured. This may occur due to:
- The use of deep markings.
- Use of an excessive volume of the coating solution.
- The air temperature is too low.
- Pan rotation speed/fluidization flow rate is too low.
Rough Surface
One of the major problems of tablet coating is the production of tablets that exhibit a rough surface. This phenomenon is often associated with the drying of the coating droplets prior to reaching the surface of the tablet. To correct this problem the spray rate may be increased and the inlet air temperature decreased.
Formation of Cracks in the Coating
The formation of cracks in tablet coatings is principally due to the use of an inappropriate coating formulation. Plasticizers are employed to lower the glass transition temperature of polymer coatings. This in turn renders the film more flexible and less brittle. Therefore cracking in polymer coatings may indicate that either the plasticizer concentration should be increased or, alternatively, a different plasticizer that is more compatible with the polymer chosen for the coating should be considered. In certain situations cracking of polymer coats may occur due to the use of a polymer that has a low-stress resistance and is therefore prone to stress failure. To rectify this situation either the molecular weight of the polymer should be increased or, alternatively, a different polymer should be used that has a greater resistance to the applied stress (i.e. an increased ultimate tensile strength).
Variations in the Colour of the Coating
Tablets that have been coated with a polymer containing a colorant should show uniform color. Variations in the color of a tablet coating may be due to:
- Improper mixing of the color within the coating formulation.
- Uneven coating process, resulting in regional differences in the thickness of the applied coating.
- Migration of colored components within the tablet core into the coating. This may be resolved by the use of a colored coating that will mask the effects of the migration or by the use of a coating in which the components within the table core are insoluble.
In-process quality control Test for Pharmaceutical Tablets
The quality in the pharmaceutical industry has become a very important and sensitive issue. Since the world has gathered together to unite its practices, guides, and the launching of the Food and Drug Administration (FDA) current good manufacturing practices (cGMP) for the 21st century – there has been a growing awareness of the significance of the quality of the pharmaceutical products.
The purposes of Quality control are to produce a perfect finished product by preventing or eliminating errors at every stage in production. Quality control is a teamwork and we have to remember that quality must be built into a drug product during product and process design and it is influenced by the physical plant design, space, ventilation, cleanliness, and sanitation during routine production.
Process testing enables the easier identification of problems.
In-process quality control (IPQC) tests were important to remove problems from every stage in production and maintain the quality of the In-process product with standards as specified in the pharmacopeias
In-Process Quality Control Tests (IPQC) are accurate and specific for testing of raw materials (RM) to the release of the finished dosage (FD) forms IPQC tests were performed in the production area.
If a defective product batch is identified, that can be corrected by rework. Whereas once that batch has been completed, this may not be possible.
Failure to meet IPC specifications indicates either those procedures were not followed or some factors were out of control. Standard operating procedures (SOPs) should be established in the pharmaceutical industry and followed that describe the IPQCs and tests:
- Size and shape: The size and shape of the tablet can be dimensionally described monitored and controlled. It is determined by the tooling during the compression process.
- Colour and odor: Many pharmaceutical tablets use color as a vital means of rapid identification and consumer acceptance. But it must be uniform within a single tablet, from tablet to tablet and from lot to lot.
The presence of an odor in a batch of tablets could indicate a stability problem e.g. the characteristic odor of acetic acid in degrading aspirin tablets or could be characteristic of the drugs e.g. vitamins have a characteristic odor. Taste is important in consumer acceptance of chewable tablets.
Size and shape: The size and shape of the tablet can be dimensionally described monitored and controlled. It is determined by the tooling during the compression process.
Colour and odor: Many pharmaceutical tablets use color as a vital means of rapid identification and consumer acceptance. But it must be uniform within a single tablet, from tablet to tablet and from lot to lot. The presence of an odor in a batch of tablets could indicate a stability problem e.g. the characteristic odor of acetic acid in degrading aspirin tablets or could be characteristic of the drugs e.g. vitamins have a characteristic odor. Taste is important in consumer acceptance of chewable tablets.
Thickness: The thickness of a tablet is the only dimensional variable related to the process. The thickness of individual tablets may be measured by a micrometer. Other techniques involve placing 5 or 10 tablets in a holding tray, where their total thickness may be measured by a sliding caliper scale. Tablet thickness should be controlled within a ± 5 % variation of a standard. Thickness must be controlled to facilitate packaging. It is expressed in mm.
Unique identification markings: Pharmaceutical companies often use some type of unique markings on tablets in addition to color, for rapid identification of their product these markings utilize some form of embossing, engraving, or printing of the company name or symbol or a product code.
The moisture content of granules: Granules should possess sufficient strength to withstand normal handling and mixing processes without breaking down and producing large amounts of fine powder. On the other hand, some size reduction during compaction into tablets is desirable to expose the areas of clean surface necessary for optimum bonding to take place so moisture content is a very important factor for producing a good pharmaceutical product.
Assay: In a tablet, an active ingredient is present which is called an Active pharmaceutical ingredient. So to prepare the tablet assay has to be done by using the suitable analytical method to produce a good finished product
Uniformity of content: A physically sound tablet may not produce the desired effects. To evaluate a tablet’s potential for efficacy, the amount of drug per tablet needs to be monitored from tablet to tablet and batch to batch. For this test according to BP using a suitable analytical method, determine the individual contents of the active substance(s) of 10 tablets taken at random. The tablet complies with the test according to BP, if each individual content is between 85% and 115% of the average content. The tablet fails to comply with the test if more than one individual contents are outside these limits or if one individual content is outside the limits of 75% to 125% of the average content. If one individual content is outside the limits of 85% to 115%, but within the limits of 75% to 125%, determine the individual contents of another 20 tablets taken at random. The tablet complies with the test if not more than one of the individual contents of the 30 tablets is outside 85% to 115% of the average content and none is outside the limits of 75% to 125% of the average content.
Uniformity of mass: This test is applicable for uncoated and film-coated tablets. For this test according to BP weigh individually 20 tablets taken at random and determine the average mass. As per BP, the tablet complies with the test if not more than 2 of the individual masses deviate from the average mass by more than the percentage deviation and none deviate by more than twice that percentage.

Weight variation test: According to the USP weight variation test is run by weighting 20 tablets individually calculating the average weights and comparing the individual tablet weights to the average. The value of the weight variation test is expressed in percentage. where Iw = Individual weight of tablet; and Aw = Average weight of the tablet.

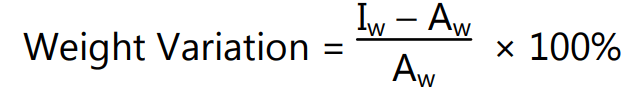
Content of Active Ingredients: For this test according to IP determine the amount of active ingredient(s) by the method described in the assay and calculate the amount of active ingredient(s) per tablet. The result lies within the range for the content of active ingredient(s) stated in the monograph.
Hardness Test: For this test one of the earliest testers was the Ketan tablet hardness tester, which is a type of the Monsanto hardness tester to evaluate tablet hardness tester. The tester consists of a barrel containing a compressible spring held between two plungers. The lower plunger is placed in contact with the tablet and zero reading is taken. The upper plunger is then forced against a spring by turning a threaded bolt until the tablet fractures. As the spring is compressed, a pointer rides along a gauge in the barrel to indicate the force. The force of fracture is recorded in kilogram.
Make sure you also check our other amazing Article on : Granulation Methods
