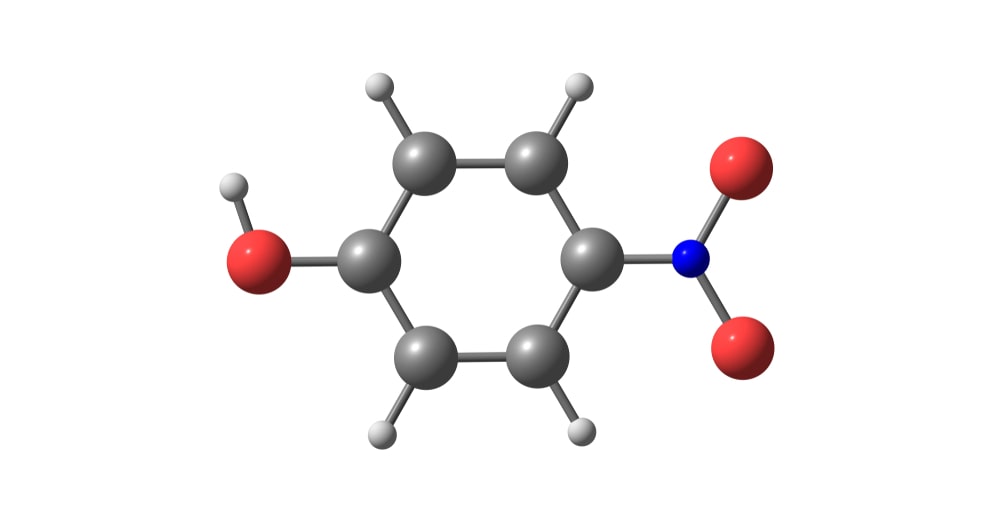
Dakin Reaction is a redox reaction in which an ortho or para-hydroxylated benzaldehyde or ketone reacts with hydrogen peroxide in alkaline conditions to form a benzenediol and a carboxylate ion.
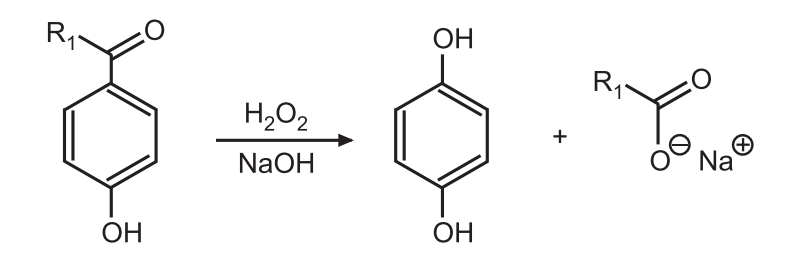
In short, the oxidation of aldehydes or ketones to the corresponding phenol is known as the Dakin reaction.
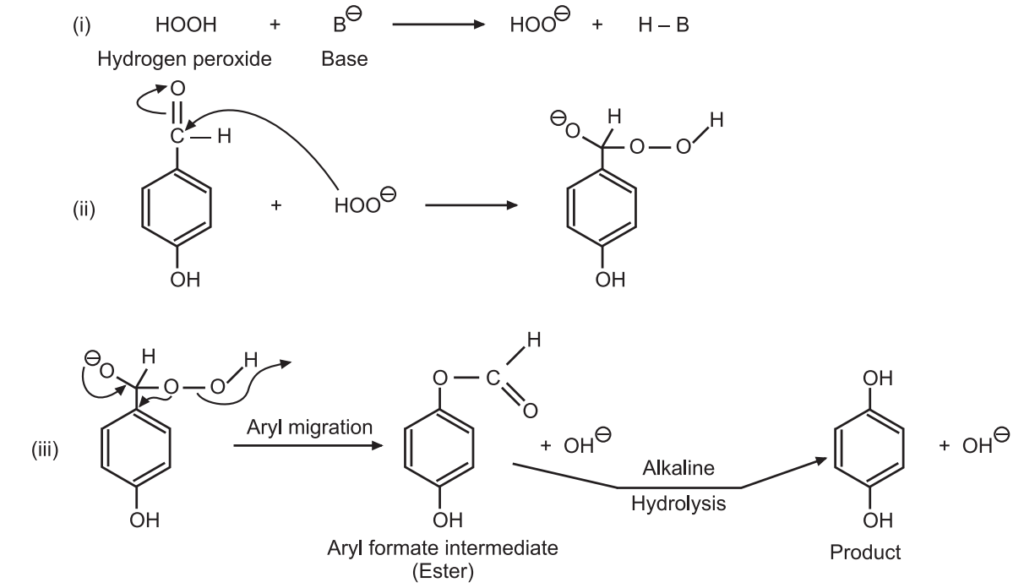
Mechanism of Dakin reaction
Check our other amazing Article on Wolff Kishner Reduction
Applications
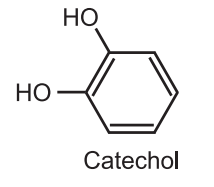
(1) Dakin reaction is used to synthesize catechol. Catechol is used as the starting material for the synthesis of several catecholamines, catecholamine derivatives, and 1, 4- tertbutyl catechol, a common antioxidant, and polymerization inhibitor.
(2) Dakin’s solution is a dilute hypochlorite solution. Chlorine, the active ingredient in Dakin’s solution, is a strong antiseptic that kills most forms of bacteria and viruses that may lead to skin and tissue infections.
Make sure you also check our other amazing Article on : Oppenauer Oxidation