Aim: To determine the Overall Heat Transfer Coefficient of water by the heat exchanger.
Requirements:
Apparatus/ Instruments: Steam Generator, Glass Condenser, Measuring Cylinder, Glass Bottle, Collecting Bowel, Thermometer, Bent tube, etc.
Principle:
When the feed of hot fluid is passed through one end of the apparatus and the cold fluid is passed through the other end, this arrangement is known as the counter-current or counter-flow method. Heat flow through fluids takes place by heat convection. The rate of heat transfer through the fluids is determined using the Overall Heat Coefficient of Liquids. i.e. the rate of heat transfer, Q=UADt
The water condenser used in the laboratory for distillation is the best example of the counter-current flow of liquids and heat transfer. Thus, the overall heat transfer coefficient is determined using a water condenser.
The overall heat transfer coefficient of a glass tube is mathematically expressed for a counter-current flow as U=Q/AD t
Where,
Q = Amount Of Heat Transferred ,W(J/S)
U=Overall heat transfer Co-Efficient W/m2.K
A= Surface Area Across Which Heat Exchange Takes Place, m2
DT= Temperature Drop
Where heat is transferred through steam to cold water through a glass surface, the total amount of heat transferred is equal to either heat lost by steam or heat gained by the water.
m1=mass of steam
t1= temperature drop between steam and water
S1= specific heat of water
L= Latent heat of vaporization of steam
The amount of heat lost by steam, (q1) =m1 s1 t1 +m1 L
Similarly, the amount of heat gained by circulating water(q2) =m2 s2 t2
Where
m2 =mass of water circulated
S2 =specific heat of water
t2 = temperature rise
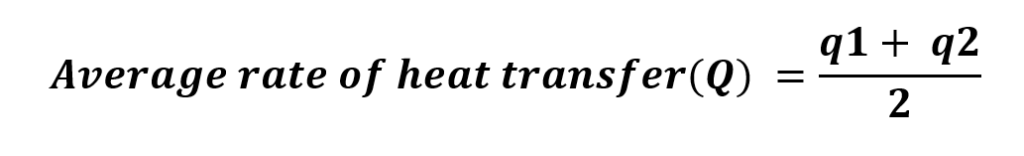
Dt in equation(1)is obtained by LMTD for counter-current flow ![]()
Applications:
- Heat exchangers are used in many industries including, refrigeration, water waste treatment, wine and beermaking, petroleum refining, etc.
- In commercial air crags,s they are used to take heat from the engine’s oil system to heat cold fuel.
- In pharmacy, it is useful in crystallization, evaporation, distillation, etc.
Procedure:
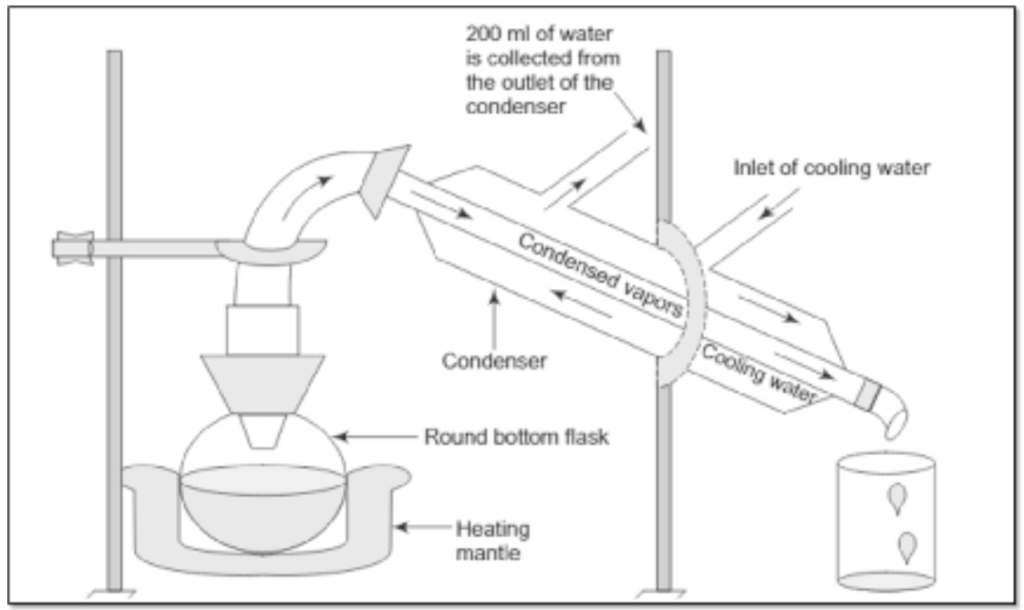
- A team generator is selected and ensured for no leakage. A sufficient quantity of water is filled in it.
- A condenser which is having the facility of circulating distillation column is selected.
- The Bend tube of the steam generator is connected to the inlet of the condenser tube.
- The outlet of the condenser tube is connected to a collecting bowl in which condensed water is to be collected.
- The circulation of cold water around the inner tube of the condenser is arranged by connecting it to a water tap.
- The outlet of the innertube of the condenser is connected to the water-collecting drum.
- Ensured that all connections are properly made before applying heat to generate steam.
- Water in the steam generator is now heated and ensured the passage of steam through the condenser tube.
- The condensation process is allowed to continue for about 5 minutes.
- Further, the water circulation is stopped from the inner tube of the condensate by stopping tap water and putting off the heating of water by means of putting off the steam generator.
- The circulating water from the exit and condensate are separately collected properly, avoiding spillage.
- The temperature of steam and condensate is noted, the difference gives the temperature drop ‘t1 ‘.
- The temperature of water at exit and entrance points are noted and the difference gives temperature rise ‘t2 ‘for the equation.
- The volume of collected condensate is measured. (m1)
- The volume of water collected is measured (m2)
- The average rate of heat transfer (Q) using the above equation is calculated and the Overall Heat transfer Co- efficiency (U) is determined.
- The entire experiment is repeated by changing the speed of the water.
- The overall Heat Transfer Coefficient for this repeated experiment is also determined.
The average rate of the Overall Heat Transfer Co-efficient is calculated by means of using the following equation.
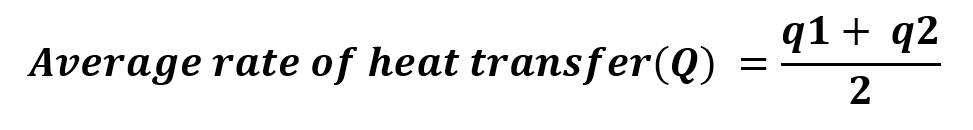
Observations
I:‐ The amount of heat LOS t by Steam“q1” IS,
“q1” =m1s1 t1+m1 L =
m1=mass of steam or condensate =
T1 = temperature of steam=
T2 = temperature of condensate =
t1 = temperature drop between steam and water =
s1 = specific heat of water = 4190 J/Kg
L =latent heat of vaporization of steam = 226.1 J/Kg
“q1” =m1s1 t1+m1 L =
II:‐Amount of heat gained by circulating water “q2” IS
“q2” =m2s2 t2
m2=mass of water circulated =
s2 = specific heat of water =
T1 = exit temperature of water =
T2 = temperature of water at inlet =
t2 =temperature rise =
“q2” = m2S2 t2
l = length of the condenser =
A = area of condenser = 2prl
r = radius of condenser
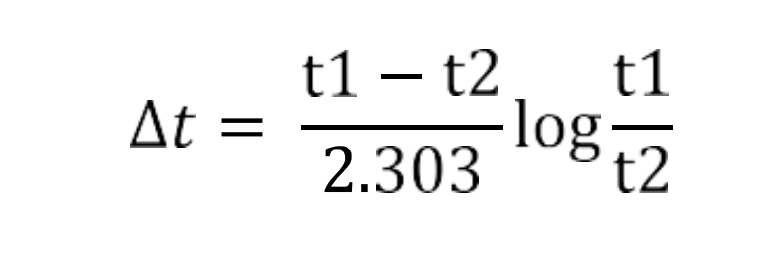
Overall heat transfers Co-efficient U=Q/AD t
RESULT:
Overall heat transfer coefficient of water “U” is = ———-
Make sure you also check our other amazing Article on: How do you calculate the efficiency of steam distillation?