Definition of pharmaceutical pellets are Small free-flowing spherical units ranging in size, prepared by agglomeration of fine powders are called pellets.
- Their size and shape allow their administration as injections and also for oral drug delivery.
- Pellets range in size, typically, between 0.5 – 1.5 mm, though other sizes could be prepared.
Table of Contents
Formulation Requirement
- Taste masking: Micropellets are ideal for products where perfect abatement of taste is required. Pellets provide the masking of unpleasant taste without lowering of bioavailability, especially for oral products.
- Immediate release: Administering drugs in pellet form leads to an increased surface area as compared to traditional compressed tablets and capsules. This would considerably reduce the time required for disintegration and have the potential for use in rapidly dispersible tablets.
- Sustained-release: Pellets are being increasingly used in the manufacture of the sustained release dosage form of drugs.
Advantages of the dosage form
- Extends daytime and nighttime activity of the drugs.
- Reduced dosage frequency of dosage forms.
- Increased patient compliance.
- Potential lower daily cost to the patient due to fewer dosage units, in contrast, the whole tablet is released at once into the small intestine as the stomach empties itself.
- Different types of polymers are utilized for the coating of different drugs to enable the sustained release/controlled release rate of drugs.
- Chemically incompatible products: At times such ingredients are required to be delivered in a single dose. In the compressed tablet dosage form separate tablets would have to be administered, but the pellets can be administered in a single capsule.
- Varying dosage without reformulation
- Pellets have excellent flow properties, due to this, they can be conveniently used for filling capsules and the manufacturer can vary the dosage by varying the capsule size without reformulating the product.
Pelletization Process and Equipment Used
Pelletization is an agglomeration process that converts fine powders or granules of bulk drugs and excipients into small, free-flowing semi-spherical units.
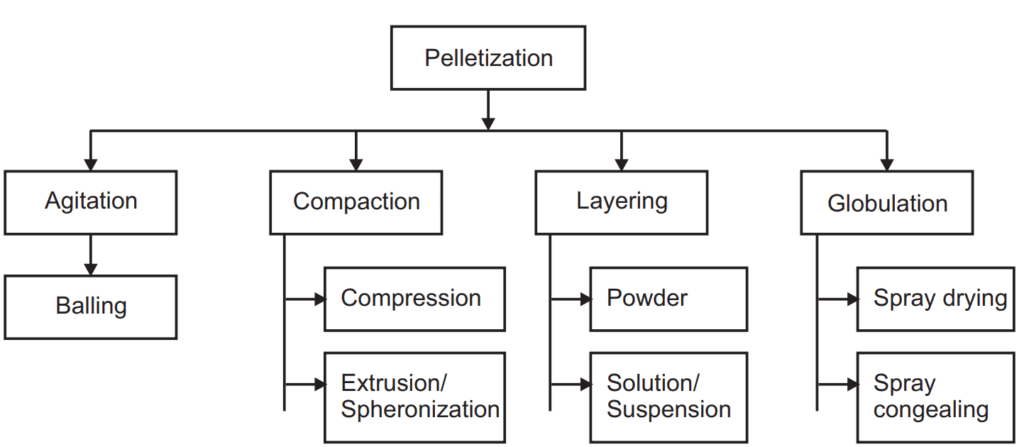
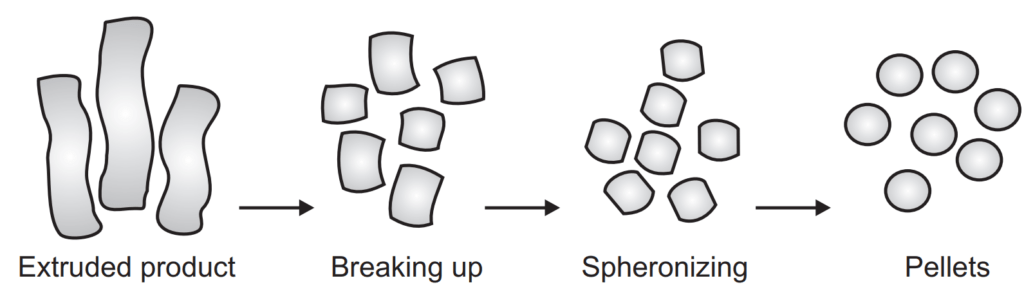
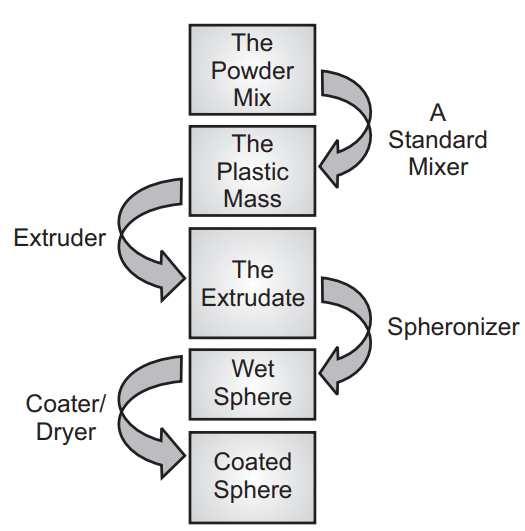
Extrusion or Spheronization
- Extrusion spheronization is widely utilized in the formulation of sustained-release, controlled-release delivery systems.
- The main objective of the extrusion spheronization is to produce pellets/spheroids of uniform size with a high drug loading capacity.
Product Features:
- Dust-free
- High sphericity
- Free-flowing
- Compact structure
- Low hygroscopicity
- High bulk density
- Low abrasion
- Narrow particle size distribution
- Smooth surface
Hot-melt extrusion
Hot melt extrusion is a process of converting raw material into a product of uniform shape and density by forcing it through a die under controlled conditions.
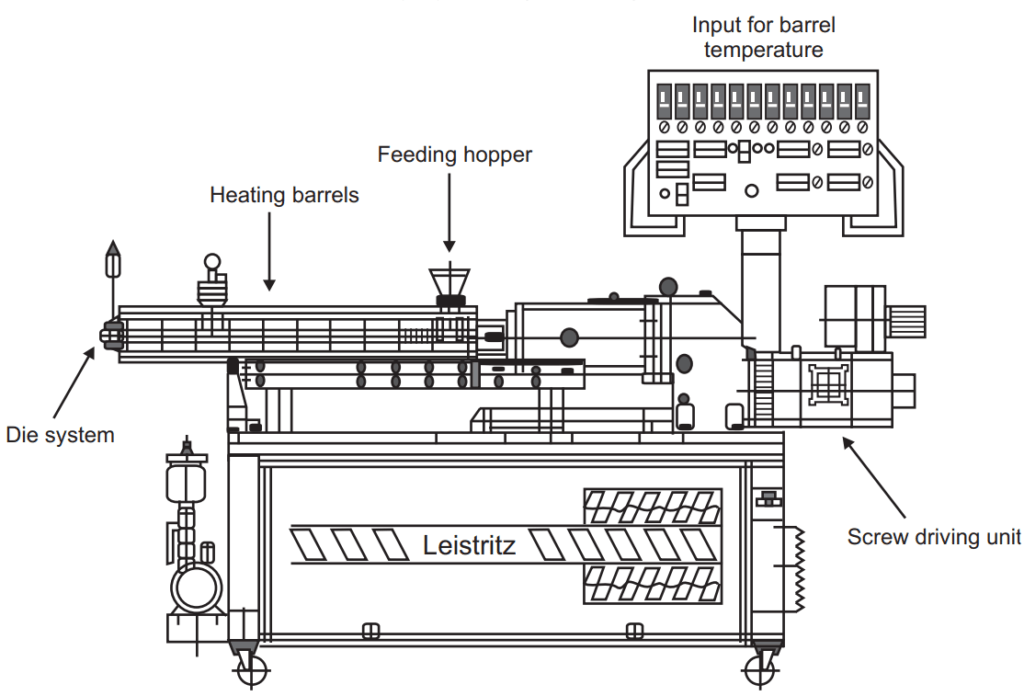
The theoretical approach to understanding the melt extrusion process is, therefore, generally presented by dividing the process of flow into four sections:
- Feeding of the extruder.
- Conveying of mass (mixing and reduction of particle size).
- Flow-through the die.
- Exit from the die and downstream processing.
Applications of Extrusion in the Pharmaceutical Industry
In the pharmaceutical industry, the melt extrusion has been used for various purposes, such as
- Improving the dissolution rate and bioavailability of the drug by forming a solid dispersion or solid solution.
- Controlling or modifying the release of the drug.
- Masking the bitter taste of an active drug.
Granulation
Wet Granulation:
- The process of adding a liquid solution to powders involves the massing of a mix of dry primary powder particles using a granulating fluid. The fluid contains a solvent that must be volatile.
- Meets all the physical requirements for compression of tablets.
Fluid-bed Granulation:
- The process is carried out continuously in a fluid-bed granulator.
- Spraying of a granulation solution onto the suspended particles, which then are dried rapidly in the hot air stream.
Fluid-Bed Granulation
The fluid-bed granulation is performed following these steps: (Tangential-Spray Method)
- Pre-blending of the formulation powder, including the active ingredients, fillers, disintegrants, in a flow of air.
- Granulation of the mixture by spraying a suitable liquid binder onto the fluidized (suspended) powder bed.
- Drying of the granulated product to the desired moisture content.
Parameters:
- Equipment parameters
- Product parameters
- Process parameters
Advantages over Traditional Wet Granulation
- Automated, performed in one unit, thus saving costs, transfer losses, and time.
- The fluid bed granulation process improves the dissolution efficiency of both nimodipine and spironolactone tablets.
Melt Granulation
Granulation is achieved by the addition of a meltable binder. Binder is in a solid state at room temperature but melts in the temperature range of 50 – 80˚C. e.g. Polyethylene Glycol (PEG) 2000, 4000, 6000, 8000 (40 – 60o C).
- The melted binder then acts as a binding liquid.
- No need for the drying phase, since dried granules are obtained by cooling them to room temperature.
Spray Drying
A drug solution or suspension is sprayed, with or without excipients, into a hot-air stream, generating dry and highly spherical particles.
Spray Congealing
Also called spray-chilling, a technique similar to spray-drying, but no source of heat is required.
- Drugs can be suspended in molten wax and can give a sustained release effect.
- Monoglycerides and similar components are spray-congealed.
Make sure you also check our other amazing Article on : Optical Rotation
