Terpenoids are the hydrocarbons of plant origin having general formula (C5H8)n. They are also oxygenated, hydrogenated and dehydrogenated derivatives. Basically, terpenoids are volatile substances which give fragrance or aroma to plants. They are also known as essential oils because they represent essence. They are mainly found in leaves and fruits of plants like conifers, citrus, and eucalyptus. The term ‘terpene’ was assigned to the compounds after the isolation of volatile liquid turpentine from pine trees. Volatile oils get vaporized or evaporate upon exposure to air at normal or ordinary temperature, so they are termed as etherel oil. Volatile oils generally occur in plant families like Umbelliferae (examples Coriander, Fennel), Zingiberaceae (Ginger), Myrtacea (Clove), Piperaceae (black pepper), Rutaceae (Lemon, Orange) etc.
Table of Contents
General properties of volatile oils or Terpenoids
- They are colourless, fragrant liquids lighter than water and volatile in nature.
- A few of them are solids e.g. camphor.
- They are soluble in organic solvents, alcohol and fixed oils and usually insoluble in water.
- They are open-chain or cyclic unsaturated compounds having one or more double bonds.
- They give an addition reaction with hydrogen, halogen, acids, etc and addition products possess antiseptic properties.
- They undergo polymerization and dehydrogenation.
- They are easily oxidized nearly by all the oxidizing agents.
- On thermal decomposition, most of the terpenoids yield isoprene as one of the products.
- They are optically active.
- It becomes darker in colour upon long-standing in air or sunlight.
- Volatile oils should be stored in well-closed airtight umbered colour containers or bottles.
Classification of Terpenoids
The terpenoids have general formula (C5H8)n. The classification is mainly done on the basis of the value of n or the number of carbon atoms present in the structure.
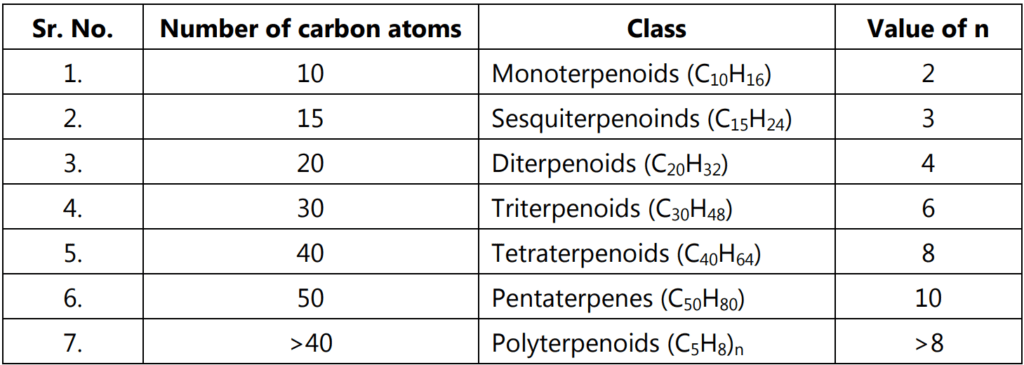
Depending upon the type and number of rings present in terpenoids, they are named as Acyclic terpenoids, Monocyclic terpenoids, Bicyclic terpenoids and Tricyclic terpenoids which contain open chain structure, one ring structure, two-ring structure and three rings structure respectively.
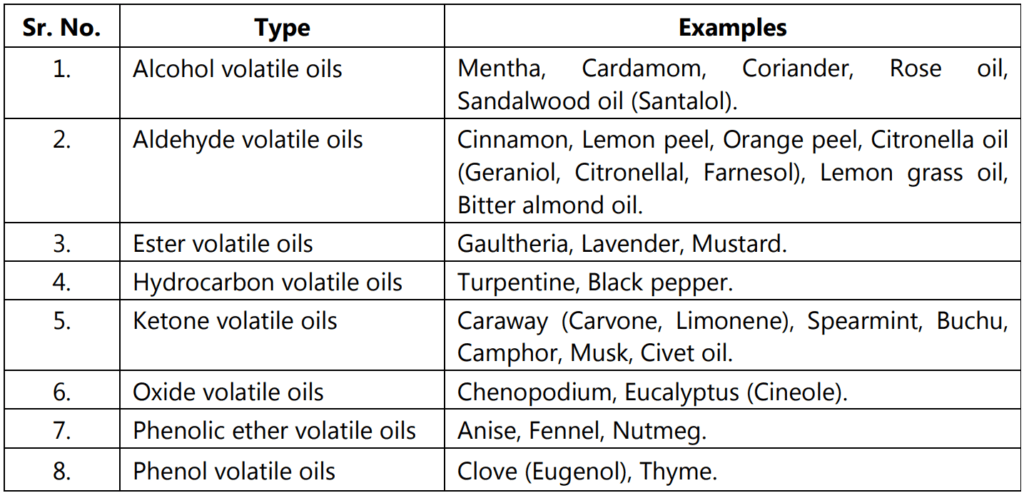
Chemical Test
- Volatile oil produces temporary stains when smeared on paper or cloth. The stain disappears after some time because the oil volatilizes.
- Upon addition of Sudan III (alcoholic solution) to the thin section of volatile oil containing drug, red colour forms.
- When a few drops of Tincture Alkana are added to a thin section of the drug, red colour develops which indicates the presence of volatile oil.
Extraction of Volatile oils
Steam distillation, solvent extraction or mechanical means like ecuelle and enfleurage methods are employed for the extraction purpose of volatile oils. These methods are as follows:
Hydro-distillation method: This method comprises of water distillation, water and steam distillation and steam distillation. These techniques are used for the extraction of volatile oils from crude drugs. Hydro-distillation technique is used for fresh leaves and steam distillation uses for subterranean parts.
Enfleurage method: This method uses for the extraction of flavouring agents and perfumery. In this method, the fresh flowers (mainly petals) are taken and spread over a layer of fatty material. Then it kept over fatty material for imbibition. After the imbibition, the fresh petals are placed on the fat layer and exhausted petals are removed. This process continues until the saturation of the fat layer with volatile constituents. This saturated fatty layer is extracted with lipid solvents.
Ecuelle method: This method is employed for citrus fruits which contains volatile principles. Here the oil containing ducts which contain oil cells are ruptured by mechanical means or manually. For this purpose, special vessels are taken which contain pointed projections. The raw material is twisted over the projections in a clockwise direction which ruptures the cell and oil comes out.
Isolation of volatile oils
The oil obtained by distillation contains a number of mono and sesquiterpenes. The monoterpenes hydrocarbons are distilled out first into their oxygenated derivatives by fractional distillation. Other methods like physical methods, chemical methods and chromatography are also used for their isolation. The chemical methods like treating the oil with sodium bisulphate, Phenylhydrazine, Tilden’s reagent and Grignard reagent isolates the compounds. Suitable adsorbents are used in chromatography for the elution of chromatograms of isolated compounds.
Uses:
The utilization of terpenoids is mainly in perfumery, cosmetic industries, soaps manufacturing, incense-sticks manufacturing, food industries, pharmaceutical industries and beverage industries.
They are also used as an antiseptic, carminative, anthelmintic, stimulant, analgesic, antirheumatic, diuretic, aromatic, counter-irritant effect, insecticide, pesticide, insect repellant, and deodorant.
Make sure you also check our other amazing Article on : Alkaloids In Pharmacognosy
